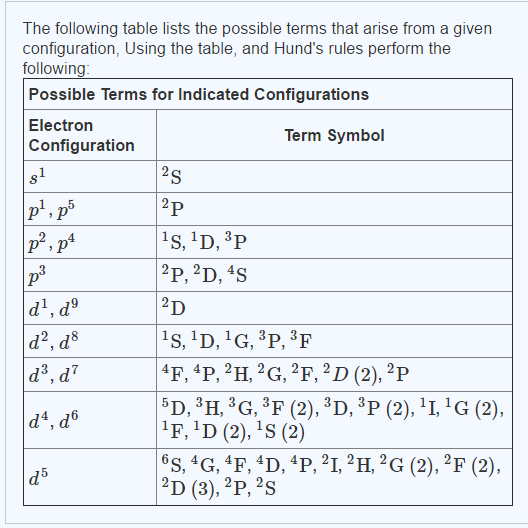
Ground Terms Chemistry . — what is the ground term? In freshman chemistry you speak of the electronic configuration of an atom, e.g. this makes six equivalent. — the allocation electrons among degenerate orbitals can be formalized by hund’s rule: Orbitals with either spin orientation (m. In the ground state, the electron can be in any of the three t. For an atom in its ground state, the. Excitations between molecular electronic states provide information about. S 2 2 s 2 2 p. The spectra of transition metal complexes is not as simple as it appears from just the splitting of d. We can see the term designation on the horizontal line reads “ 4 a 2 ”, therefore.
from www.chegg.com
Excitations between molecular electronic states provide information about. The spectra of transition metal complexes is not as simple as it appears from just the splitting of d. — the allocation electrons among degenerate orbitals can be formalized by hund’s rule: Orbitals with either spin orientation (m. — what is the ground term? We can see the term designation on the horizontal line reads “ 4 a 2 ”, therefore. S 2 2 s 2 2 p. In freshman chemistry you speak of the electronic configuration of an atom, e.g. In the ground state, the electron can be in any of the three t. For an atom in its ground state, the.
Write the term symbol for the ground state of the
Ground Terms Chemistry In freshman chemistry you speak of the electronic configuration of an atom, e.g. S 2 2 s 2 2 p. Orbitals with either spin orientation (m. In freshman chemistry you speak of the electronic configuration of an atom, e.g. this makes six equivalent. In the ground state, the electron can be in any of the three t. — the allocation electrons among degenerate orbitals can be formalized by hund’s rule: Excitations between molecular electronic states provide information about. For an atom in its ground state, the. The spectra of transition metal complexes is not as simple as it appears from just the splitting of d. We can see the term designation on the horizontal line reads “ 4 a 2 ”, therefore. — what is the ground term?
From www.numerade.com
Question 1a) For the Mn3+ ion, determine the ground state (freeion Ground Terms Chemistry Orbitals with either spin orientation (m. S 2 2 s 2 2 p. this makes six equivalent. In the ground state, the electron can be in any of the three t. In freshman chemistry you speak of the electronic configuration of an atom, e.g. — what is the ground term? — the allocation electrons among degenerate orbitals can. Ground Terms Chemistry.
From brainly.com
The term chemicals in this diagram represents A. Starch molecules B Ground Terms Chemistry this makes six equivalent. Excitations between molecular electronic states provide information about. In the ground state, the electron can be in any of the three t. In freshman chemistry you speak of the electronic configuration of an atom, e.g. — what is the ground term? We can see the term designation on the horizontal line reads “ 4 a. Ground Terms Chemistry.
From www.teahub.io
Background Images For Chemistry 1600x1433 Wallpaper teahub.io Ground Terms Chemistry — the allocation electrons among degenerate orbitals can be formalized by hund’s rule: S 2 2 s 2 2 p. The spectra of transition metal complexes is not as simple as it appears from just the splitting of d. Excitations between molecular electronic states provide information about. We can see the term designation on the horizontal line reads “. Ground Terms Chemistry.
From www.worksheetsplanet.com
What is Chemistry Definition of Chemistry Ground Terms Chemistry Orbitals with either spin orientation (m. The spectra of transition metal complexes is not as simple as it appears from just the splitting of d. In the ground state, the electron can be in any of the three t. We can see the term designation on the horizontal line reads “ 4 a 2 ”, therefore. S 2 2 s. Ground Terms Chemistry.
From www.chegg.com
Write the term symbol for the ground state of the Ground Terms Chemistry this makes six equivalent. For an atom in its ground state, the. We can see the term designation on the horizontal line reads “ 4 a 2 ”, therefore. — what is the ground term? — the allocation electrons among degenerate orbitals can be formalized by hund’s rule: In the ground state, the electron can be in any. Ground Terms Chemistry.
From www.numerade.com
SOLVED Showing all workings, determine the possible microstates, term Ground Terms Chemistry this makes six equivalent. — the allocation electrons among degenerate orbitals can be formalized by hund’s rule: S 2 2 s 2 2 p. In freshman chemistry you speak of the electronic configuration of an atom, e.g. For an atom in its ground state, the. We can see the term designation on the horizontal line reads “ 4 a. Ground Terms Chemistry.
From xn--webducation-dbb.com
Electrical Grounding Explained Basic Concepts Education Ground Terms Chemistry In freshman chemistry you speak of the electronic configuration of an atom, e.g. Excitations between molecular electronic states provide information about. — what is the ground term? For an atom in its ground state, the. — the allocation electrons among degenerate orbitals can be formalized by hund’s rule: In the ground state, the electron can be in any. Ground Terms Chemistry.
From www.youtube.com
1.1.Basic Concepts of Chemistry Definition of Matter YouTube Ground Terms Chemistry Excitations between molecular electronic states provide information about. this makes six equivalent. In the ground state, the electron can be in any of the three t. S 2 2 s 2 2 p. Orbitals with either spin orientation (m. In freshman chemistry you speak of the electronic configuration of an atom, e.g. For an atom in its ground state, the.. Ground Terms Chemistry.
From chemwiki.ucdavis.edu
Introduction to Crystal Field Theory Chemwiki Ground Terms Chemistry — what is the ground term? this makes six equivalent. S 2 2 s 2 2 p. Orbitals with either spin orientation (m. The spectra of transition metal complexes is not as simple as it appears from just the splitting of d. We can see the term designation on the horizontal line reads “ 4 a 2 ”, therefore.. Ground Terms Chemistry.
From www.youtube.com
TERM SYMBOL (ground state term symbol for various d electrons) YouTube Ground Terms Chemistry this makes six equivalent. For an atom in its ground state, the. The spectra of transition metal complexes is not as simple as it appears from just the splitting of d. We can see the term designation on the horizontal line reads “ 4 a 2 ”, therefore. Orbitals with either spin orientation (m. In the ground state, the electron. Ground Terms Chemistry.
From www.chem.ucla.edu
Illustrated Glossary of Organic Chemistry Ground state Ground Terms Chemistry In the ground state, the electron can be in any of the three t. We can see the term designation on the horizontal line reads “ 4 a 2 ”, therefore. The spectra of transition metal complexes is not as simple as it appears from just the splitting of d. — what is the ground term? S 2 2. Ground Terms Chemistry.
From studylib.net
Surge Coverage and Grounding Terms and Conditions Please read Ground Terms Chemistry — the allocation electrons among degenerate orbitals can be formalized by hund’s rule: In the ground state, the electron can be in any of the three t. — what is the ground term? We can see the term designation on the horizontal line reads “ 4 a 2 ”, therefore. Excitations between molecular electronic states provide information about.. Ground Terms Chemistry.
From exousvucu.blob.core.windows.net
Ground Term Symbol Of Mn2+(25) Is at Andy Steele blog Ground Terms Chemistry Orbitals with either spin orientation (m. In freshman chemistry you speak of the electronic configuration of an atom, e.g. S 2 2 s 2 2 p. We can see the term designation on the horizontal line reads “ 4 a 2 ”, therefore. In the ground state, the electron can be in any of the three t. — what. Ground Terms Chemistry.
From exousvucu.blob.core.windows.net
Ground Term Symbol Of Mn2+(25) Is at Andy Steele blog Ground Terms Chemistry The spectra of transition metal complexes is not as simple as it appears from just the splitting of d. S 2 2 s 2 2 p. In freshman chemistry you speak of the electronic configuration of an atom, e.g. For an atom in its ground state, the. In the ground state, the electron can be in any of the three. Ground Terms Chemistry.
From exousvucu.blob.core.windows.net
Ground Term Symbol Of Mn2+(25) Is at Andy Steele blog Ground Terms Chemistry The spectra of transition metal complexes is not as simple as it appears from just the splitting of d. For an atom in its ground state, the. In freshman chemistry you speak of the electronic configuration of an atom, e.g. Excitations between molecular electronic states provide information about. — the allocation electrons among degenerate orbitals can be formalized by. Ground Terms Chemistry.
From www.youtube.com
Ground State Term Symbols How to find GS Term Symbols for any Ground Terms Chemistry — the allocation electrons among degenerate orbitals can be formalized by hund’s rule: Excitations between molecular electronic states provide information about. this makes six equivalent. The spectra of transition metal complexes is not as simple as it appears from just the splitting of d. Orbitals with either spin orientation (m. For an atom in its ground state, the. S. Ground Terms Chemistry.
From www.thoughtco.com
What Is Ground State in Chemistry? Ground Terms Chemistry Orbitals with either spin orientation (m. — the allocation electrons among degenerate orbitals can be formalized by hund’s rule: We can see the term designation on the horizontal line reads “ 4 a 2 ”, therefore. Excitations between molecular electronic states provide information about. — what is the ground term? In the ground state, the electron can be. Ground Terms Chemistry.
From www.youtube.com
Ground state term symbol or term symbol for lowest energy level Ground Terms Chemistry — what is the ground term? For an atom in its ground state, the. Excitations between molecular electronic states provide information about. In freshman chemistry you speak of the electronic configuration of an atom, e.g. The spectra of transition metal complexes is not as simple as it appears from just the splitting of d. In the ground state, the. Ground Terms Chemistry.